|
1/1/2024 0 Comments Ap chem periodic table 
Particles is going to be proportional, that just means We can view Coulomb's Law as saying that the magnitude of the force between two charged In chemistry or physics, and that's Coulomb's Law. And to do so, we're going to start with a very fundamental idea We're going to look at trends for the periodic table of elements for dimensions like ionization energy, atomic and ionic radii, electron affinity, and electro negativity. Noble gas notation is not saying that a noble gas like argon has 18 core electrons, rather that it has a complete valence shell. So calcium's electron configuration can also be written as: Argon's electron configuration is: 1s^(2)2s^(2)2p^(6)3s^(2)3p^(6), which we can condense by saying. So writing calcium's electron configuration would look like:ġs^(2)2s^(2)2p^(6)3s^(2)3p^(6)4s^(2), normally.īut with noble gas notation we can use the noble gas prior to calcium, argon, as a foundation. The idea is that noble gases have completed shells so elements can build off the electron configurations of previous noble gases to express their own electrons configuration. The previous video talked about using noble gas notation to write the electron configurations of higher atomic number elements. It breaks down even more when you get to the heavier noble gases and you have to worry about the f electrons too. So it's no longer simple the previous noble gas's (Argon) atomic number because of the d electrons. The reason your rule begins to break down at krypton is because the filling of the d subshell adds an extra 10 electrons to the core electrons. Krypton's valence electrons are the 4s and 4p electrons (again in total eight) which means the remaining 28 electrons are core electrons. 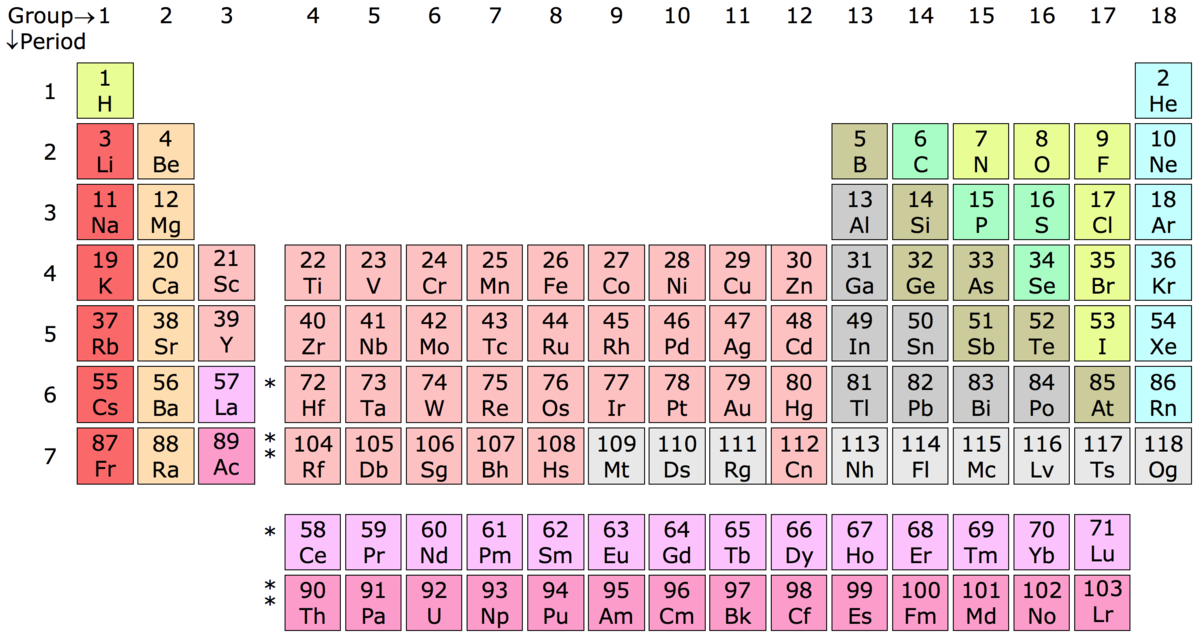
So your rule does work, but only for noble gases up to argon. 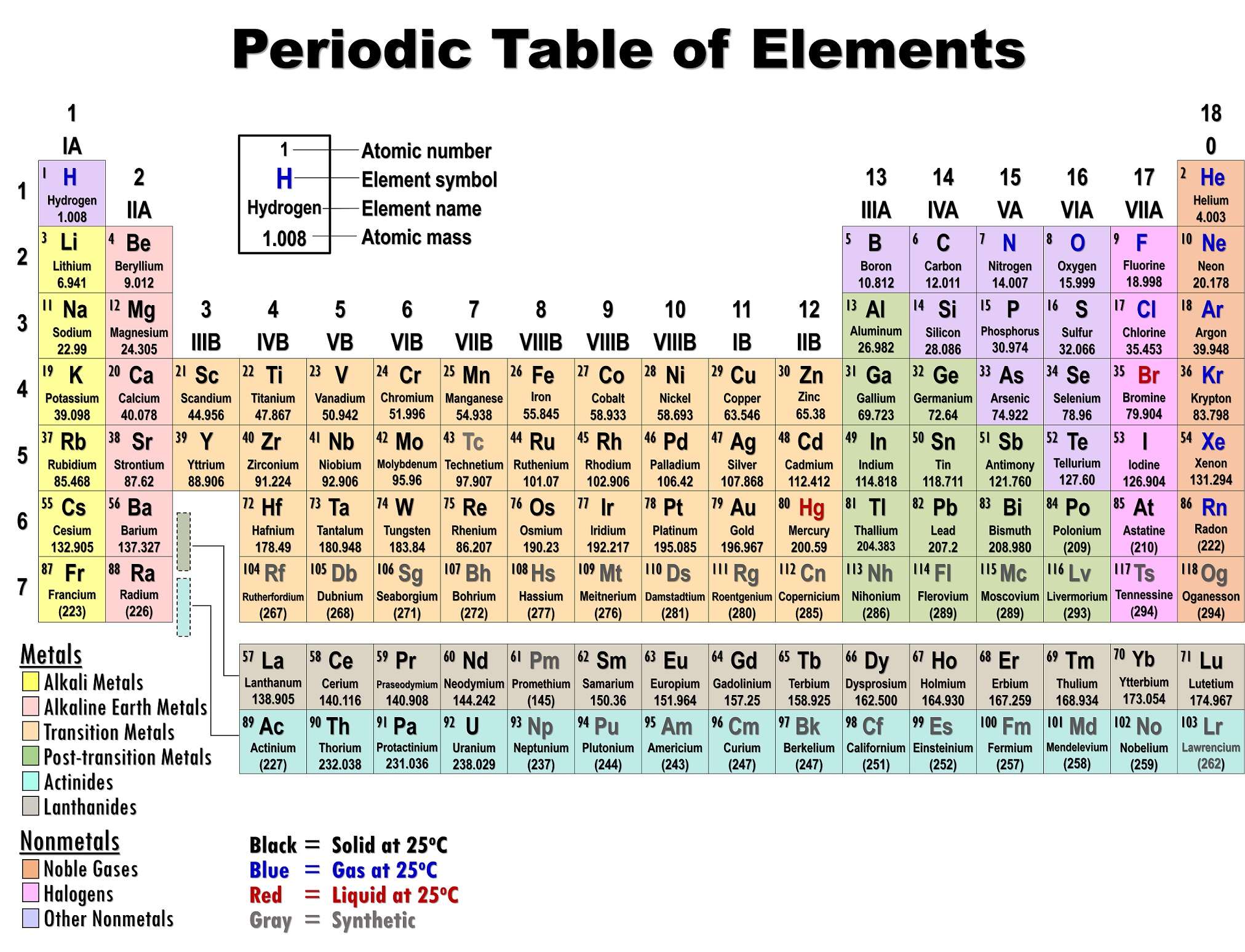
Neon's valence electrons are the 2s and 2p electrons (in total eight) and the two 1s electrons in the lower shell are the core electrons.Īrgon also has eight valence electrons, but this time the 3s and 3p electrons meaning the remaining ten in the lower first and seconds shells are core electrons. There isn't a shell below the first electron shell so it has no core electrons. All other electrons would be considered core electrons.įor helium it only has two 1s electrons which constitute the valence shell. For noble gases the valence electrons are the s and p electrons of the highest electron shell. The core electrons for any atom are all the electrons of the atom which aren't valence electrons. Hope that starts to clarify things a little more. Electrons are always partially in the nucleus. All electron states overlap with the nucleus, so the concept of an electron "crashing into" the nucleus does not really make sense. The states with more energy are more spread out. An electron in an atom spreads out according to its energy. In fact, electrons in the s orbitals tend to peak at the nucleus. And using this understanding of the electron, electrons in the atom do enter the nucleus. Under quantum mechanics the electron is a quantized wave function which occupies certain probable regions around the nucleus called orbitals. To accurately describe the nature of electrons, we must resort to quantum mechanics. This is using classical mechanics to describe electrons when in reality they behave much more differently. As always, you have the flexibility to organize the course content as you like.So the issue with thinking electrons could crash into the nucleus is that it assumes electrons are solid particles orbiting the nucleus according to Coulomb's Law much like how planets orbit the sun because of gravity. The AP Chemistry framework is organized into nine commonly taught units of study that provide one possible sequence for the course. The framework also encourages instruction that prepares students for advanced chemistry coursework. The framework specifies what students must know, be able to do, and understand, with a focus on big ideas that encompass core principles and theories of the discipline. Based on the Understanding by Design® (Wiggins and McTighe) model, this course framework provides a clear and detailed description of the course requirements necessary for student success. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
